3: The Properties of Oxygen Gas (Experiment)
- Page ID
- 93984
- To generate (and collect) oxygen gas via the decomposition of hydrogen peroxide.
- To investigate the properties of oxygen, particularly as an agent of combustion.
Oxygen is one of the most abundant elements on this planet. Our atmosphere is 21% free elemental oxygen. Oxygen is also extensively combined in compounds in the earths crust, such as water (89%) and in mineral oxides. Even the human body is 65% oxygen by mass.
Free elemental oxygen occurs naturally as a gas in the form of diatomic molecules, \(\ce{O2}\) (g). Oxygen exhibits many unique physical and chemical properties. For example, oxygen is a colorless and odorless gas, with a density greater than that of air, and a very low solubility in water. In fact, the latter two properties greatly facilitate the collection of oxygen in this lab. Among the unique chemical properties of oxygen are its ability to support respiration in plants and animals, and its ability to support combustion.
In this lab, oxygen will be generated as a product of the decomposition of hydrogen peroxide. A catalyst is used to speed up the rate of the decomposition reaction (without being consumed by the reaction), which would otherwise be too slow to use as a source of oxygen. The particular catalyst used here is active yeast.
The reaction for generating oxygen gas from hydrogen peroxide is shown below:
\[\ce{2 H2O2 (aq) ->[catalyst] 2 H2O (l) + O2 (g)}\]
\[\text{hydrogen peroxide} \ce{->[catalyst]} \text{water} + \text{oxygen}\]
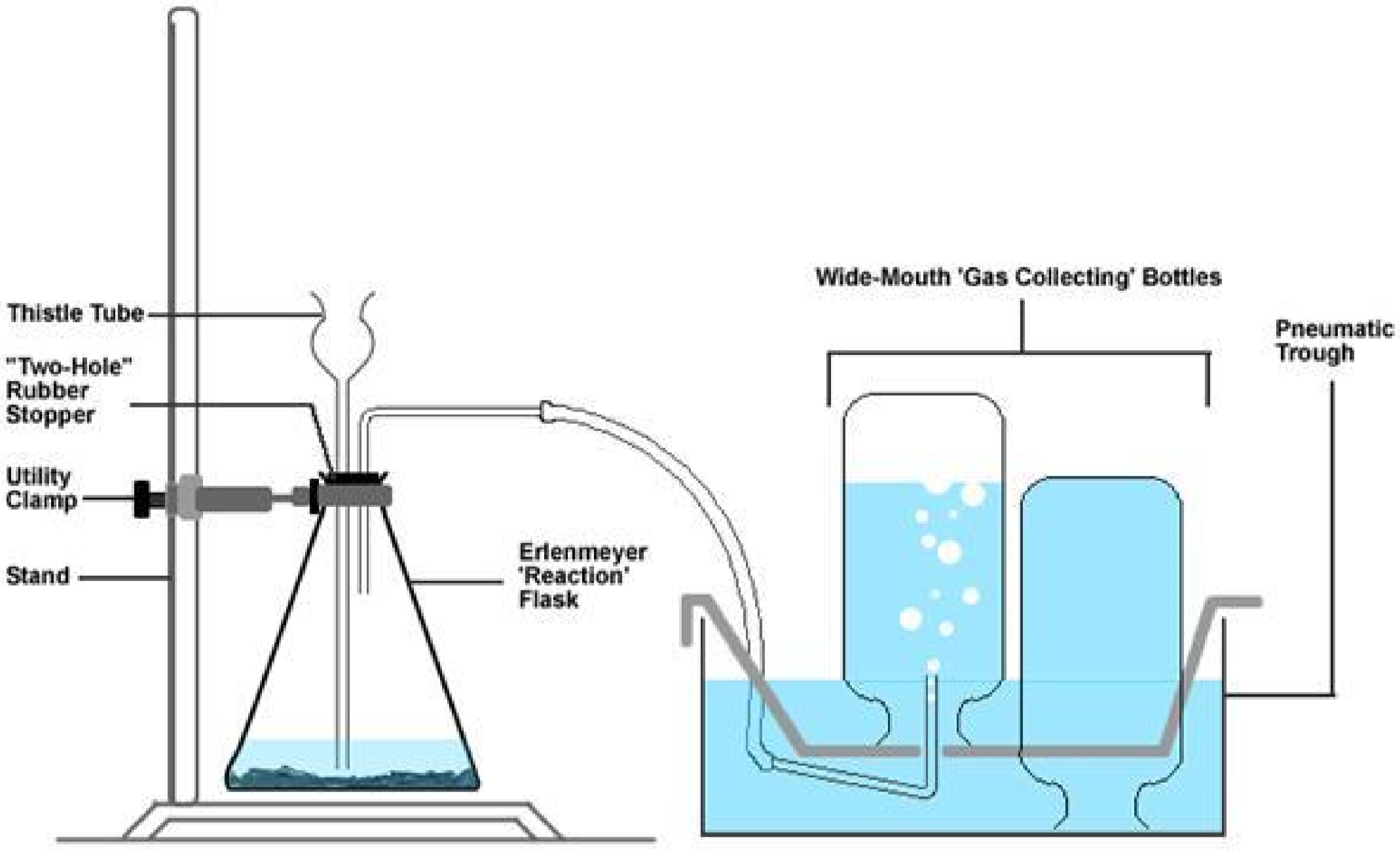
The oxygen gas produced will be collected in bottles by a method known as the downward displacement of water (see figure below). Once collected, several tests will be performed in order to investigate the role of oxygen in a variety of combustion reactions.
A combustion reaction is commonly referred to as “burning”. During a combustion reaction, oxygen reacts chemically with the substance being burned. Note that since our atmosphere is roughly 21% oxygen, many substances readily burn in air. Both oxygen and the substance being burned (the reactants) are consumed during the combustion reaction, while new substances (the products) and heat energy are generated. Since heat is produced, this is an exothermic reaction.
Combustion reactions can be described by the following chemical equation:
\[\text{Substance being burned} + \text{Oxygen} \ce{->} \text{Products} + \text{Heat}\]
The actual products of a combustion reaction depend on what substance is burned and how much oxygen is present. In general, however, when a pure element burns in oxygen the product is called an oxide. An oxide is a compound containing both the element and oxygen chemically combined together.
Some examples of element combustion are shown below. Several such reactions will be performed using the oxygen gas collected in this lab.
Combustion of an Element can be described using the following chemical equation:
\[\text{Element} + \text{Oxygen} \ce{->} \text{Oxide of Element} + \text{Heat}\]
Two examples are:
\[\ce{C (s) + O2 (g) -> CO2(g) +} \text{Heat}\]
\[\text{Carbon} + \text{Oxygen} \ce{->} \text{Carbon Dioxide} + \text{Heat}\]
\[\ce{2 Hg (l) + O2 (g) -> 2 HgO (s) +} \text{Heat}\]
\[\text{Mercury} + \text{Oxygen} \ce{->} \text{Mercury (II) Oxide} + \text{Heat}\]
Procedure
Materials and Equipment
Materials: hydrogen peroxide solution, active yeast, wooden splints, candle, sulfur, steel wool, magnesium ribbon, aluminum pellets, 6 M hydrochloric acid
Equipment: 250-mL Erlenmeyer flask, five wide-mouth bottles, four glass ‘cover’ plates, pneumatic trough, “stopper + thistle tube + tubing” apparatus, utility clamp, ring stand, deflagration spoon, crucible tongs, small beaker, medium test tube and plastic test tube rack.
Exercise caution and/or use gloves when using the hydrogen peroxide (\(\ce{H2O2}\)) and the hydrochloric acid (\(\ce{HCl}\)) as they can cause chemical burns and skin irritation. If either of these chemicals comes into contact with your skin, immediately rinse with water for a minimum of fifteen minutes and notify your instructor.
Also, do not look directly at the burning magnesium. In addition to being very bright, it emits harmful UV radiation that could cause damage to the retina of the eye.
Personal protective equipment (PPE) required: lab coat, safety goggles, closed-toe shoes, gloves
Part A: Generating and Collecting Oxygen Gas
- Obtain the following equipment:
- 250-mL Erlenmeyer flask (locker)
- “two-hole stopper + thistle tube + glass tubing + rubber tubing” apparatus (stockroom)
- five wide mouth ‘gas-collecting’ bottles (under sink)
- four glass ‘cover’ plates (front desk)
- pneumatic trough (under sink) filled with water to 1⁄2 inch above the metal shelf
- Fill four of the five wide-mouth bottles to the brim with water (the fifth will be used later). Then gently slide a glass plate over the mouth of each bottle. Make sure that there are no air bubbles at the top of the glass plate.
- While holding the glass plate, gently invert a bottle and lower it into the water in the trough. Remove the glass plate when the mouth of the bottle is below the water level in the trough. Repeat this for all four bottles. Place the glass plates aside, as they will be used later.
- Place one gas-collecting bottle on the metal shelf. Make sure that the mouth of the bottle is below the water level.
- Now focus on your reaction vessel, the Erlenmeyer flask. Add a pea-sized amount of active yeast (the catalyst) to the flask, followed by approximately 25-mL of distilled water.
- Finally, assemble all your equipment together as demonstrated by your instructor, or as shown in the figure below. Make sure that
- the end of the thistle tube is completely covered with water at the bottom of the flask,
- the end of the glass tubing running from the Erlenmeyer flask is inserted under the opening in the bottom of the metal shelf into the gas-collecting bottle (which is full of water),
- the Erlenmeyer flask is stabilized with a utility clamp
- Obtain about 30-mL of hydrogen peroxide (\(\ce{H2O2}\)) in your smallest beaker. Then carefully add about 10-mL of this \(\ce{H2O2}\) through the thistle tube. The generation of oxygen gas should begin immediately. If at any time the rate of the reaction in the Erlenmeyer flask appears to slow down, add another 10-mL portion of \(\ce{H2O2}\).
- The oxygen produced will fill the inverted bottle by displacing the water in it. This is because oxygen does not dissolve in water, due to its low solubility. When the first bottle is completely filled with gas, place the second bottle on the metal rack in its place and allow it to fill in a like manner. Repeat this for the third and fourth bottles.
- As soon as each bottle is completely filled, remove it by placing a glass plate under the bottle’s mouth while under water, then lifting the bottle and plate from the pneumatic trough. Place the bottle on the lab bench mouth up and do not remove the glass plate. Since oxygen is denser than air, it sinks to the bottom of the flask and will not readily leak out the top.
- Using masking tape, label each bottle of gas in the order they are collected: Bottle #1, Bottle #2, Bottle #3 and Bottle #4. Label the fifth unused bottle “Empty Bottle”.
- Once all four bottles are filled with oxygen, do not add any more \(\ce{H2O2}\) to the Erlenmeyer flask. Set it aside and allow the reaction to go to completion. At the end of the lab, the chemicals remaining in the reaction flask and any unused \(\ce{H2O2}\) must be disposed of in the labeled container in the hood. In the meantime, proceed to Part B.
Part B: The Properties of Oxygen Gas
Test 1: Combustion of wood
Light a wooden split, and then blow it out. While it is still glowing, place it in the empty wide- mouth bottle (air-filled only). Record your observations. Now re-light the same wooden split, and again blow it out. Quickly insert the splint into Bottle #1 (oxygen-filled) while it is still glowing. Record your observations.
Test 2: Combustion of candle wax
Place a tealight candle on a glass plate and light it. Lower the empty bottle over the candle. Measure and record the number of seconds that the candle continues to burn. Then re-light the candle and lower Bottle #2 over it. Again, measure and record the number of seconds that the candle continues to burn.
Test 3: Combustion of sulfur
This test must be performed in the hood. Take the empty bottle and Bottle #3 to the hood your instructor directs you to. Place a small lump of sulfur in a deflagrating spoon. Light the Bunsen burner in the hood, and heat the sulfur in the spoon. The sulfur will first melt, then burn with an almost invisible blue flame. Insert the spoon with the burning sulfur in the empty bottle and record your observations. Then insert it in Bottle #3, and again record your observations. When finished, extinguish the burning sulfur in a beaker of water.
Test 4: Combustion of iron
Pour about 30-mL of tap water into Bottle #4 and replace the glass plate quickly. Take a loose (frayed out) 2 or 3 centimeter piece of steel wool and hold it in a Bunsen burner flame for a very brief instant with your crucible tongs (it will glow red). Then immediately lower the steel wool into Bottle #4. Record your observations. Repeat with the empty bottle and record your observations.
Test 5: Combustion of hydrogen
This test must be performed in air only. (Note: The hydrogen burned in this test must be first generated by a reaction between aluminum and hydrochloric acid.) To a medium test tube, add aluminum pellets, followed by about 3-mL of hydrochloric acid. Bubbles should begin to appear as hydrogen gas is produced. Place the test tube in the plastic test tube rack. After 30-60 seconds have elapsed, light a wooden splint. Do not blow it out. Hold the burning splint to the mouth of test tube (where the hydrogen gas is being evolved) and record your observations.
Test 6: Combustion of magnesium
This test is an instructor demonstration. It must be performed in air only. Hold a 1-inch piece of magnesium metal in a Bunsen burner flame with your crucible tongs until it ignites (in air). Record your observations, remembering not to look directly at the burning magnesium!
Lab Report: The Properties of Oxygen Gas
Part A: Generating and Collecting Oxygen Gas
- Write the equation for the reaction used to generate oxygen gas.
- Word Equation:
- Balanced Chemical Equation:
- What is the name of the catalyst used in this reaction?
- What is the purpose of this catalyst? (There are two parts to this answer.)
- In addition to oxygen, what other substance is produced by this reaction? What happens to this substance?
- Two notable physical properties of oxygen are its low solubility in water and a density greater than air.
- Which one of these properties allows the oxygen gas to be collected via the displacement of water? Explain.
- Which one of these properties allows the oxygen gas collected to be stored in the bottles mouth up? Explain.
- What percentage of the air we breathe is oxygen gas?
Part B: The Properties of Oxygen Gas
| Test | Observations |
|---|---|
|
Test 1 |
Observations |
|
Glowing splint in empty bottle |
|
|
Glowing splint in Bottle #1 |
|
|
Test 2 |
Observations |
|
Burning candle in empty bottle |
|
|
Burning candle in Bottle #2 |
|
|
Test 3 |
Observations |
|
Burning sulfur in empty bottle |
|
|
Burning sulfur in Bottle #3 |
|
|
Test 4 |
Observations |
|
Glowing steel in empty bottle |
|
|
Glowing steel in Bottle #4 |
|
|
Test 5 |
Observations |
|
Burning hydrogen in air |
|
|
Test 6 |
Observations |
|
Burning magnesium in air |
Analysis of Combustion Results
- Why did you perform each test in two separate bottles (air-filled and oxygen-filled)?
- Are the combustion reactions of oxygen exothermic or endothermic? Support your answer with experimental evidence from the tests you performed.
- Consider your results for the first four tests you performed. In which bottles, air-filled or oxygen-filled, did the combustion reactions occur more vigorously? Why?
- Consider your Test 2 results. Although the candle burns for a longer period of time in one bottle, it eventually goes out in both the empty bottle and Bottle #2. Why does it go out?
- When an element burns in oxygen gas, the product is called an oxide.
- The wood in the splint consists mostly of carbon. The combustion of carbon produces carbon dioxide, \(\ce{CO2}\). Write the equation for the combustion of wood (carbon).
- Balanced Chemical Equation:
- The combustion of sulfur produces sulfur dioxide, \(\ce{SO2}\). Write the equation for the combustion of sulfur.
- Balanced Chemical Equation:
- The combustion of hydrogen produces water, \(\ce{H2O}\). Write the equation for the combustion of hydrogen.
- Balanced Chemical Equation:
- Steel wool consists mostly of iron. The combustion of iron produces iron(III) oxide, \(\ce{Fe2O3}\). Write the equation for the combustion of steel wool (iron).
- Balanced Chemical Equation: