Cisplatin 8. Discovery of Cisplatin
- Page ID
- 2905
As we saw in the role of platinum electrodes module, the researchers found that certain transition metal complexes—specifically, soluble metal salts—caused elongation of E. coli bacterial cells. Among them was the ammonium salt of platinum hexachloride ((NH4)2PtCl6), in which platinum existed in its +4 oxidation state. Upon closer examination, however, the researchers realized that light was necessary for the filamentous growth of the bacteria to occur. In fact, exposure of this soluble platinum salt to ultraviolet light produced a new, neutral species:
(NH4)2Pt(IV)Cl6 + uv light --> Pt(IV)(NH3)2Cl4
This neutral product was chemically identical to the electrolytically formed agent and was shown to cause bacterial elongation. However, when one of the chemists in the group did a control experiment in which he synthesized this new, neutral species, diamminetetrachloroplatinum(IV) (PtIV(NH3)2Cl4), by known chemical techniques, he found that no bacterial elongation occurred!1 In order to explain this surprising result, the researchers thought more carefully about the compounds and realized that there were, in fact, two possible structures with the molecular formula PtIV(NH3)2Cl4, as shown in Figure 1.
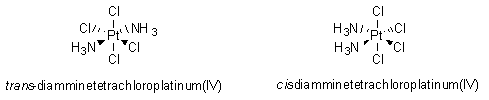
In the structure on the left, the two amino (NH3) groups are on opposite sides of the central platinum(IV) atom; we say that the amino groups are trans to each other. In the structure on the right, the two amino groups are on the same side of the molecule, or cis to each other. (see the transition metal chemistry module) The trans configuration is more thermodynamically stable than the cis conformation, and the trans molecule was the one that the researchers first synthesized and studied. When they made the compound on the right, with the cis configuration, they achieved complete success.1 Along with different possible geometries of transition metal complexes, the central transition metals can have different possible oxidation states. (See the redox chemistry module.) For example, platinum has two dominant oxidation states, +2 and +4. The studies described above were done with platinum complexes in which platinum was in the +4 oxidation state; such complexes are octahedral, as can be seen above with the trans- and cis- diamminetetrachloroplatinum(IV) complexes. On the other hand, when platinum is in the +2 oxidation state, it forms square planar complexes. The researchers decided to synthesize and test a variety of neutral platinum complexes, having platinum with either the +2 or +4 oxidation state. Among others, they made the following four compounds: (1) cis-PtIV(NH3)2Cl4; (2) cis-PtII(NH3)2Cl2; (3) PtII(NH2CH2CH2NH2)Cl2; and (4) PtIV(NH2CH2CH2NH2)Cl4, as shown in Figure 2.
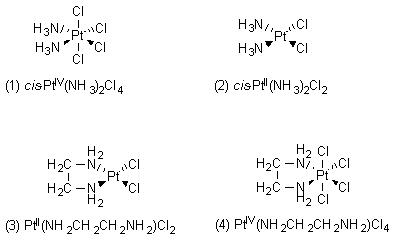
Having shown that these four compounds inhibited bacterial cell division without being toxic to these cells, the researchers also wanted to see how these compounds would affect cell division in mammalian cells. They thought that perhaps the platinum-containing compounds would stop cell division in tumor cells without being extremely toxic to the host animal. For this reason they turned their attention to two murine (mouse) cancer cell lines, Sarcoma 180 and Leukemia L1210.1,2 First, the researchers had to determine safe dose levels for mice. One of the biologists found that the dose that killed half of the animals (LD50, meaning "lethal dose, 50%") was about 13 mg of the drug per kilogram of animal body weight (in other words, LD50 = 13 mg/kg). A nonlethal dose for mice was 8 mg/kg.1 The next step was to see whether these compounds could be used to treat murine tumors without killing the mouse. At this point, another biologist implanted a standard transplantable animal tumor (solid Sarcoma 180) beneath the skin under one armpit of each mouse. Tumor growth was determined by cutting out the tumor and weighing it; this could be done because the tumor was nonmetastatic, meaning that it remained localized under the armpit of the mouse. After 10 days, the tumor had increased to 100 times its original mass.1 Different conditions were used for the experiments that followed. The first set of experiments followed the standard protocol of the National Cancer Institute. After implantation of the tumor on day 0, the drug was injected on day 1, and the animals were sacrificed on day 8. The efficacy of the drug was determined by finding the ratio of the tumor size in the treated group to that in the control group (T/C). When this ratio was less than 0.5, the drug was considered to be effective in treating the tumor. The biologist conducted many experiments on the mice, dosing them with different nonlethal amounts of the platinum-containing compounds shown above. The results showed that all of these drugs reduced the size of the tumors and that their T/C ratios were all well below 0.5. Compound 2 (cis-PtII(NH3)2Cl2, or cis-diamminedichloroplatinum(II)) was the most effective of all the compounds in reducing the size of the tumors, as it had the lowest T/C ratios.1,2 In another set of experiments, the tumors were implanted into the mice on day 0, and instead of injecting the drugs on day 1, the tumors were allowed to grow for 8 days until they reached the mass of a gram; this was a fairly sizable tumor, considering that the mice only weighed about 20 grams each (see photo, Figure 3). On day 8 the test group of mice was injected with one of the platinum-containing compounds. Again, compound 2 (cis-PtII(NH3)2Cl2, or cis-diamminedichloroplatinum(II)) gave dramatic results, as shown in Figure 3.
Figure 3. Time sequence photographs of two mice with solid Sarcoma 180 tumors. The mouse at the top was an untreated negative control. She died on day 21 when the tumor weighed about 3 g. The bottom mouse was in the group treated on day 8 with an intraperitoneal injection of cis-diamminedichloroplatinum(II). Her tumor was completely regressed six days after treatment, and she died of age-related causes almost 3 years later. Reprinted with permission.1
References
- Rosenberg, B. In Nucleic Acid-Metal Ion Interactions. T. G. Spiro, Ed. John Wiley & Sons, Inc.: New York, 1980, Vol. 1, pp. 1-29.
- Rosenberg, B., VanCamp, L., Trosko, J. E., Mansour, V. H. Nature,1969, 222, pp. 385-386.



