The Effect of Temperature on the NO2/N2O4 Equilibrium
- Page ID
- 3073
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
Chemical Concept Demonstrated
- Temperature's effect on equilibrium
Demonstration
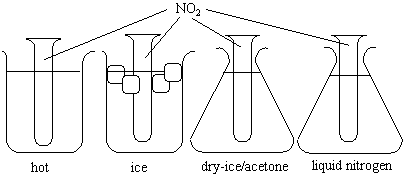
Four glass tubes are sealed with NO2 gas.
|
 |
Observations
NO2 is brown and N2O4 is colorless. The intensity of the brown color decreases as the temperature decreases. Therefore, a decrease in temperature yields and increase in N2O4. Equilibrium is shifted to the N2O4 side upon a decrease in temperature.
Explanation (including important chemical equations)
2 NO2 (g) <=> N2O4 (g)
The standard enthalpy (delta H° = -57.2 kJ) and the entropy (delta S° = -175.83 kJ) of reaction can be calculated from the follow standard-state enthalpies of formation and standard-state entropies.
| Compound | delta H°f (kJ/mol) | delta S° (J/mol-K) |
| NO2 | 33.18 | 240.06 |
| N2O4 | 9.16 | 304.29 |
We can then invoke the assumption that the temperature dependence of delta H° for this reaction is small to estimate the equilibrium constant at various temperatures.
| Temperature | delta G° (kJ) | Kp |
| 100 °C | 8.4 | 0.066 |
| 0 °C | -9.2 | 58 |
| -78 °C | -22.9 | 1.3 x 106 |
| -196 °C | -43.6 | 3.7 x 1029 |
Contributors
- Dr. George Bodner (Perdue University)

