Le Chatelier's Principle - NO₂/N₂O₄ Equilibrium
- Page ID
- 3008
Chemical Concept Demonstrated
- LeChatelier's Principle
Demonstration
| Two glass tubes are filled with gas, one with NO2 and one with NO and NO2.
Both of these tubes are immersed in liquid nitrogen. As an alternative, fill two clear sealed syringes with NO2 gas, place both on an overhead projector, and press one of the plungers in. |
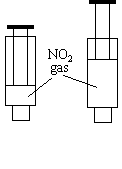 |
Observations
The glass tube with NO and NO2 in it produces a blue liquid. The flask with NO2 in it produces a white solid. If the syringes are used, the compressed gas first becomes darker, then becomes lighter than the control syringe as the plunger is depressed.
Explanation
NO and NO2 in the same tube are in equilibrium with the compound N2O3, which is a blue liquid. At room temperature, the equilibrium is shifted far to the end of the gases. However, when the temperature is lowered, LeChatelier's Principle comes into play. Because the formation of N2O3 is exothermic, lowering the temperature of the system makes the reaction more favorable.
A similar application of the principle explains why pure NO2 forms N2O4 (a white powder) at low temperatures.
In the syringe, the gas becomes darker at first simply because the gas is compressed into a smaller space. The gas lightens because the resulting system is not in equilibrium. Because different gases take up the same space with the same number of particles, it is advantageous for two NO2 molecules to form a single molecule under high-pressure conditions. N2O4 is produced, fewer particles are in the syringe, and, as a result, the intensity of the color caused by the NO2 goes down.
Contributors
- Dr. George Bodner (Perdue University)

