Catalysts
- Page ID
- 9623
A catalyst is a substance that speeds up a reaction without being consumed by it. More specifically, a catalyst provides an alternative, lower activation energy pathway between reactants and products. As such, catalysts are vitally important to chemical technology; approximately 95% of industrial chemical processes involve catalysts of various kinds. In addition, most biochemical processes that occur in living organisms are mediated by enzymes, which are catalysts made of proteins.
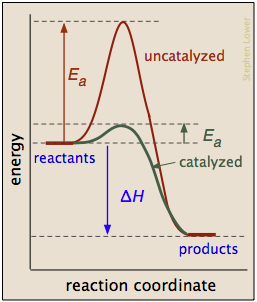
It is important to understand that a catalyst affects only the kinetics of a reaction; it does not alter the thermodynamic tendency of the reaction to occur. Therefore, ΔHis the same for the two pathways depicted in the plot above.
Contributors and Attributions
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook

