Conformations of Biphenyls
- Page ID
- 1255
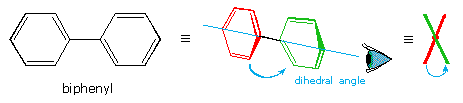
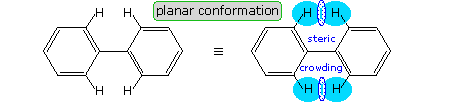
Another class of compounds that display conformational enantiomorphism are the substituted biphenyls. As shown in the following diagram, biphenyl itself is not planar, one benzene ring being slightly twisted or canted in relation to the other as a consequence of steric crowding. This crowding will be demonstrated by clicking on the diagram. The resulting chiral conformation, having a dihedral angle of about 45º, equilibrates rapidly with its enantiomer by rotation about the connecting single bond. Note that a conformation having a 90º dihedral angle is achiral, as a consequence of a plane of symmetry.
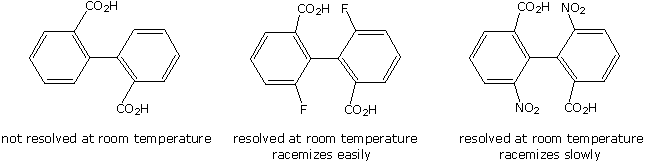
If each of the phenyl rings of a biphenyl has two different ortho or meta substituents (one may be hydrogen), even the twisted 90º dihedral angle conformer becomes chiral. In order to interconvert such conformers with their mirror image structures, a rotation through the higher energy coplanar form must be made. The ease with which this interconversion occurs will depend on the size of the ortho substituents, since these groups must slide past each other. The 2,2'-dicarboxylic acid on the left below cannot be resolved at room temperature, since thermal (kinetic) energy is sufficient to provide the necessary activation energy for racemization. The two additionally substituted diacids to its right have a higher activation energy for racemization, and can be resolved if care is taken to avoid heating them. As a rule, an activation energy barrier of 16 to 19 kcal/mole is required to prevent spontaneous room temperature racemization of substituted biphenyls. Since fluorine is smaller than a nitro group, the center compound racemizes more rapidly on heating than does the nitro compound to its right. Conformational isomers that are isolable due to high energy barriers are called atropisomers.
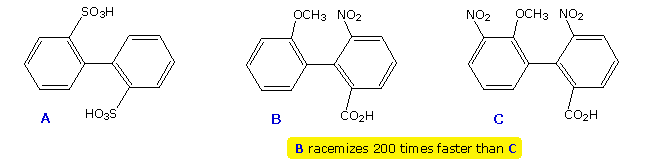
The 2,2'-disulfonic acid (compound A) can be resolved with care, confirming the larger size of SO3H compared with CO2H. Compounds B and C provide additional insight into the racemization of biphenyls. Although these biphenyls have identical ortho substituents, the meta nitro substituent adjacent to the methoxyl group in C exerts a buttressing influence that increases the effective size of that ortho substituent.
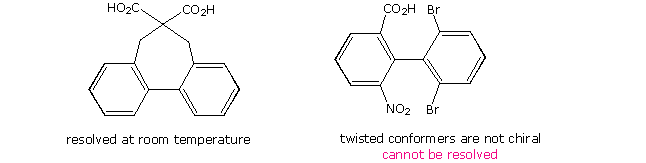
Finally, by clicking on the diagram a second time two additional examples of substituted biphenyls will be shown. The left hand compound is held in a twisted conformation by the bridging carbon chain. Racemization requires passing through a planar configuration, and the increased angle and eclipsing strain in this structure contribute to a large activation energy. Consequently, this compound is easily resolved into enantiomeric stereoisomers. The right hand compound is heavily ortho-substituted and most certainly resists assuming a planar configuration. However, the right benzene ring has two identical ortho substituents, so the stable 90º dihedral angle conformer has a plane of symmetry. All chiral twisted conformers are present as racemates, so this compound cannot be resolved.
To see models of biphenyl and a chiral tetrasubstituted derivative .
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


