Hydrogenation of Alkenes
- Page ID
- 898
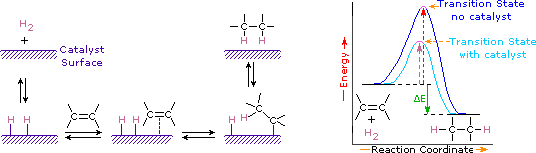
Addition of hydrogen to a carbon-carbon double bond is called hydrogenation. The overall effect of such an addition is the reductive removal of the double bond functional group. Regioselectivity is not an issue, since the same group (a hydrogen atom) is bonded to each of the double bond carbons. The simplest source of two hydrogen atoms is molecular hydrogen (H2), but mixing alkenes with hydrogen does not result in any discernible reaction. Although the overall hydrogenation reaction is exothermic, a high activation energy prevents it from taking place under normal conditions. This restriction may be circumvented by the use of a catalyst, as shown in the following diagram.
Catalysts are substances that changes the rate (velocity) of a chemical reaction without being consumed or appearing as part of the product. Catalysts act by lowering the activation energy of reactions, but they do not change the relative potential energy of the reactants and products. Finely divided metals, such as platinum, palladium and nickel, are among the most widely used hydrogenation catalysts. Catalytic hydrogenation takes place in at least two stages, as depicted in the diagram. First, the alkene must be adsorbed on the surface of the catalyst along with some of the hydrogen. Next, two hydrogens shift from the metal surface to the carbons of the double bond, and the resulting saturated hydrocarbon, which is more weakly adsorbed, leaves the catalyst surface. The exact nature and timing of the last events is not well understood.
As shown in the energy diagram, the hydrogenation of alkenes is exothermic, and heat is released corresponding to the ΔE (colored green) in the diagram. This heat of reaction can be used to evaluate the thermodynamic stability of alkenes having different numbers of alkyl substituents on the double bond. For example, the following table lists the heats of hydrogenation for three C5H10 alkenes which give the same alkane product (2-methylbutane). Since a large heat of reaction indicates a high energy reactant, these heats are inversely proportional to the stabilities of the alkene isomers. To a rough approximation, we see that each alkyl substituent on a double bond stabilizes this functional group by a bit more than 1 kcal/mole.
| Alkene Isomer | (CH3)2CHCH=CH2 3-methyl-1-butene |
CH2=C(CH3)CH2CH3 2-methyl-1-butene |
(CH3)2C=CHCH3 2-methyl-2-butene |
|---|---|---|---|
| Heat of Reaction ( ΔHº ) |
–30.3 kcal/mole | –28.5 kcal/mole | –26.9 kcal/mole |
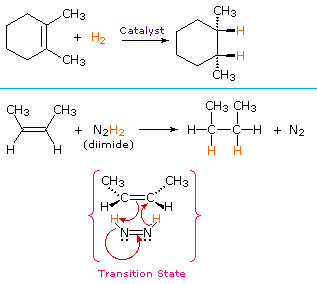
From the mechanism shown here we would expect the addition of hydrogen to occur with syn-stereoselectivity. This is often true, but the hydrogenation catalysts may also cause isomerization of the double bond prior to hydrogen addition, in which case stereoselectivity may be uncertain.
The formation of transition metal complexes with alkenes has been convincingly demonstrated by the isolation of stable platinum complexes such as Zeise's salt, K[PtCl3(C2H4)].H2O, and ethylenebis(triphenylphosphine)platinum, [(C6H5)3P]2Pt(H2C=CH2). In the latter, platinum is three-coordinate and zero-valent, whereas Zeise's salt is a derivative of platinum(II). A model of Zeise's salt and a discussion of the unusual bonding in such complexes may be viewed by clicking here. Similar complexes have been reported for nickel and palladium, metals which also function as catalysts for alkene hydrogenation.
A non-catalytic procedure for the syn-addition of hydrogen makes use of the unstable compound diimide, N2H2. This reagent must be freshly generated in the reaction system, usually by oxidation of hydrazine, and the strongly exothermic reaction is favored by the elimination of nitrogen gas (a very stable compound). Diimide may exist as cis-trans isomers; only the cis-isomer serves as a reducing agent. Examples of alkene reductions by both procedures are shown below.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry