Reactions of Group I Elements with Oxygen
- Page ID
- 3671
- To understand the reactions of the Group 1 elements (lithium, sodium, potassium, rubidium and cesium) with oxygen
- To understand the simple reactions of the various oxides
This page examines the reactions of the Group 1 elements (lithium, sodium, potassium, rubidium and cesium) with oxygen, and the simple reactions of the various oxides formed.
The Reactions with Oxygen
Group 1 metals are very reactive, and must be stored out of contact with air to prevent oxidation. Reactivity increases as you go down the group; the less reactive metals (lithium, sodium and potassium) are stored in oil (because of its density, lithium floats in oil, but because it is less reactive than the other metals in the group, the thin coating of oil that results is sufficient to prevent reaction).

Sodium metal stored under oil. .
Rubidium and cesium are typically stored in sealed glass tubes to eliminate contact with air. They are stored either in a vacuum or in an inert gas such as argon and the tubes must be broken open when the metal is used.

Rubidium metal sample from the Dennis s.k collection. .
Depending on the period of the metal, a different type of oxide is formed when the metal is burned . The reactions are the same in oxygen and in air, but oxygen will generate a more violent reaction. Lithium is unique in the group because it also reacts with the nitrogen in the air to form lithium nitride.
Lithium
Lithium burns with a strongly red-tinged flame if heated in air; in pure oxygen, the flame is more intense. It reacts with oxygen in the air to give white lithium oxide:
\[ 4Li + O_2 \rightarrow 2Li_2O \label{1} \]
Lithium also reacts with the nitrogen in the air to produce lithium nitride and is the only Group 1 element that forms a nitride:
\[ 6Li + N_2 \rightarrow 2Li_3N \label{2} \]
Sodium
Small pieces of sodium burn in air with a faint orange glow. Using larger amounts of sodium or burning it in oxygen gives a strong orange flame. The reaction produces a white solid mixture of sodium oxide and sodium peroxide. The equation for the formation of the simple oxide is analogous to the lithium equation:
\[ 4Na + O_2 \rightarrow 2Na_2O \label{3} \]
The peroxide equation is:
\[ 2Na + O_2 \rightarrow Na_2O_2 \label{4} \]
Potassium
Small pieces of potassium heated in air melt and convert instantly into a mixture of potassium peroxide and potassium superoxide without a visible flame. Larger pieces of potassium produce a lilac flame. The equation for the formation of the peroxide is like the sodium equation above:
\[ 2K + O_2 \rightarrow K_2O_2 \label{5} \]
The superoxide reaction is as follows:
\[ K + O_2 \rightarrow KO_2 \label{6} \]
Rubidium and Cesium
Both rubidium and cesium metals ignite in air and produce superoxides, \(RbO_2\) and \(CsO_2\). The equations for these reactions are analogous to the equivalent potassium superoxide equation (Equation 6):
\[ Rb + O_2 \rightarrow RbO_2 \label{7} \]
\[ Cs + O_2 \rightarrow CsO_2 \label{8} \]
Both superoxides are described as either orange or yellow, but rubidium superoxide can also be dark brown.
Why are different oxides formed as you go down the Group?
The oxide forms of each element can be summarized as follows:
- Lithium (and to some extent sodium) form simple oxides, \(X_2O\), which contain the common \(O^{2-}\) ion .
- Sodium (and to some extent potassium) form peroxides, \(X_2O_2\), containing the more complicated \(O_2^{2-}\) ion.
- Potassium, rubidium and cesium form superoxides, \(XO_2\). containing the superoxide ion, \(O_2^-\).
The more complicated ions are unstable in the presence of a small positive ion. Consider the peroxide ion, \(O_2^{2-}\), which has the following structure:

The covalent bond between the two oxygen atoms is relatively weak. Now imagine bringing a small positive ion close to the peroxide ion. Electrons in the peroxide ion will be strongly attracted toward the positive ion. A simple oxide ion can be formed if the oxygen atom on the right "breaks off":
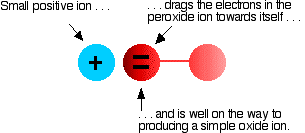
Hence, the positive ion polarizes the negative ion. This is most effective if the positive ion is small and highly charged (if it has a high charge density, or a lot of charge packed into a small volume).
Even though it has only a +1 charge, the lithium ion at the top of the group is very small small; therefore it has a high enough charge density that any peroxide ion near it breaks down into an oxide and an oxygen atom. Larger Group 1 ions have less of an effect on the peroxide ion because of their low charge density. The superoxide (\(O_2^-\)) ions are even more easily pulled apart; these are only stable in the presence of the large ions toward the bottom of the group.
The larger metals form complicated oxides due to energetic factors. In the presence of sufficient oxygen, the compound which produces the most stable compound is dominant (Table 1).
| \(\Delta H \;(kJ/mol\; of\; Rb)\) | |
|---|---|
| Rb2O | -169.5 |
| Rb2O2 | -236 |
| RbO2 | -278.7 |
The values for the various potassium oxides show the same trends. As long as there is enough oxygen, forming the peroxide releases more energy per mole of metal than forming the simple oxide. Forming the superoxide has an even greater enthalpy change.
Reactivity of the simple Oxides: \(X_2O\)
- Reaction of Oxides with water: Simple metal oxides are basic, reacting with water to produce the metal hydroxide. For example, lithium oxide reacts with water to give a colorless solution of lithium hydroxide:
\[ X_2O + H_2O \rightarrow 2X^+_{(aq)} + OH^-_{(aq)} \label{9} \]
- Reaction of Oxides with dilute acids: Simple metal oxides react with acids, producing a salt and water. For example, sodium oxide reacts with dilute hydrochloric acid to give colorless sodium chloride solution and water:
\[ X_2O + 2HCl \rightarrow 2XCl + H_2O \label{10} \]
Reactivity of the Peroxides: \(X_2O_2\)
- Reaction of Peroxides with water: If the reaction is takes place under ice-cold conditions (and the temperature is controlled to be constant despite the exothermicity of these reactions), then a solution of the metal hydroxide and hydrogen peroxide is formed: \[ X_2O_2 + 2H_2O \rightarrow 2XOH + H_2O_2 \label{11} \] If the temperature increases (as it inevitably does unless the peroxide is added to water very slowly), the hydrogen peroxide produced decomposes into water and oxygen: \[2H_2O_{2(l)} \rightarrow 2H_2O_{(l)} + O_{2(g)} \label{12} \] This reaction can be extremely violent .
- Reaction of Peroxides with dilute acids: These reactions are even more exothermic than those with water. A solution containing a salt and hydrogen peroxide is formed. Hydrogen peroxide decomposes to water and oxygen if the temperature rises, an almost unavoidable event. This reaction, given below, is also potentially violent. \[ X_2O_2 + 2HCl \rightarrow 2XCl + H_2O_2 \label{13} \]
Reactivity of the Superoxides: \(XO_2\)
- Reaction of Superoxides with water: In this reaction, a solution of the metal hydroxide and hydrogen peroxide is formed, but oxygen gas is also generated. Once again, these are strongly exothermic reactions and the heat produced inevitably decomposes the hydrogen peroxide to water and more oxygen.
\[ 2XO_2 + 2H_2O \rightarrow 2XOH + H_2O_2 + O_2 \label{14} \]
- Reaction of Superoxides with dilute acids: These reactions are again more exothermic and violent than those with water. A solution containing a salt and hydrogen peroxide is formed together with oxygen gas. The hydrogen peroxide decomposes to water and oxygen as the temperature rises.
\[ 2XO_2 + 2HCl \rightarrow 2XCl + H_2O_2 + O_2 \label{15} \]
Summary
Forming complicated oxides from the metals releases more energy and makes the system more energetically stable. However, this only applies to the lower half of the group, in which the metal ions are large and have a low charge density. At the top of the group, the small ions with a higher charge density tend to polarize the more complicated oxide ions to the point of disintegration.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


