Chemical Reactions Overview
- Page ID
- 235
Chemical reactions are the processes by which chemicals interact to form new chemicals with different compositions. Simply stated, a chemical reaction is the process where reactants are transformed into products. How chemicals react is dictated by the chemical properties of the element or compound- the ways in which a compound or element undergoes changes in composition.
Describing Reactions Quantitatively
Chemical reactions are constantly occurring in the world around us; everything from the rusting of an iron fence to the metabolic pathways of a human cell are all examples of chemical reactions. Chemistry is an attempt to classify and better understand these reactions.
A chemical reaction is typically represented by a chemical equation, which represents the change from reactants to products. The left hand side of the equation represents the reactants, while the right hand side represents the products. A typical chemical reaction is written with stoichiometric coefficients, which show the relative amounts of products and reactants involved in the reaction. Each compound is followed by a parenthetical note of the compound’s state of 2: (l) for liquid, (s) for solid, (g) for gas. The symbol (aq) is also commonly used in order to represent an aqueous solution, in which compounds are dissolved in water. A reaction might take the following form:
\[\ce{A (aq) + B (g) \rightarrow C (s) + D (l)} \nonumber \]
In the above example, \(A\) and \(B\), known as the reactants, reacted to form \(C\) and \(D\), the products.
To write an accurate chemical equation, two things must occur:
- Each product and reactant must be written using its chemical formula, e.g., \(H_2\)
- The number of atoms of each element must be equal on both sides of the equation. Coefficients are used in front of the chemical formulas in order to help balance the number of atoms, e.g.,
\[\ce{2Mg + O_2 \rightarrow 2MgO} \nonumber \]
Hydrogen and nitrogen react together in order to produce ammonia gas, write the chemical equation of this reaction.
Solution
Step 1: Write each product and reactant using its chemical formula.
\[\ce{H_2 + N_2 \rightarrow NH_3} \nonumber \]
Step 2: Ensure the number of atoms of each element are equal on both sides of the equation.
\[\ce{3H_2 + N_2 \rightarrow 2NH_3} \nonumber \]
In order to balance this equation, coefficients must be used. Since there are only 2 nitrogen atoms present on the left side of the equation, a coefficient of 2 must be added to \(NH_3\).
Stoichiometry
The coefficient that used for balancing the equation is called the stoichiometric coefficient. The coefficients tell us the ratio of each element in a chemical equation. For example
\[\ce{2Mg + O_2 \rightarrow 2MgO} \nonumber \]
means
- 2 moles of MgO is produced for every 2 moles of Mg consumed.
- 2 moles of MgO is produced for every 1 mole of O2 consumed.
When all of the reactants of a reaction are completely consumed, the reaction is in perfect stoichiometric proportions. Often times, however, a reaction is not in perfect stoichiometric proportions, leading to a situation in which the entirety of one reactant is consumed, but there is some of another reactant remaining. The reactant that is entirely consumed is called the limiting reactant, and it determines how much of the products are produced.
4.00 g of hydrogen gas mixed with 20.0g of oxygen gas. How many grams of water are produced?
Solution
\[n(H_2)=\dfrac{4g}{(1.008 \times2)g/mol}=1.98mol \nonumber \]
So theoretically, it requires 0.99 mol of \(O_2\)
n(O2)=n(H2)*(1mol O2/2mol H2)=0.99 mol
m(O2)=n(O2)*(16g/mol *2) = 31.7 g O2
Because \(O_2\) only has 20.0 g, less than the required mass. It is limiting.
Often, reactants do not react completely, resulting in a smaller amount of product formed than anticipated. The amount of product expected to be formed from the chemical equation is called the theoretical yield. The amount of product that is produced during a reaction is the actual yield. To determine the percent yield:
Percent yield =actual yield/theoretical yield X 100%
Chemical reactions do not only happen in the air, but also exist in solutions. In a solution, the solvent is the compound that is dissolved, and the solute is the compound that the solvent is dissolved in. The molarity of a solution is the number of moles of a solvent divided by the number of liters of solution.
\[\ Molarity=\dfrac{ \text{amount of solute (mol)}}{\text{volume of solution (L)}} \nonumber \]
\[\ M=\dfrac{n}{V} \nonumber \]
100.0 g NaCl is dissolved in 50.00 ml water. What is the molarity of the solution?
Solution
a) Find the amount of solute in moles.
100.0g/(22.99 g/mol+35.45 g/mol) =1.711 moles
b) Convert mL to L.
50.00 mL=0.05000 L
c) Find the molarity
1.711 moles/0.05000L=34.22 mol/L
Physical Changes During Chemical Reactions
Physical change is the change in physical properties. Physical changes usually occur during chemical reactions, but does not change the nature of substances. The most common physical changes during reactions are the change of color, scent and evolution of gas. However, when physical changes occur, chemical reactions may not occur.
Types of Chemical Reactions
Precipitation, or double-replacement reaction
A reaction that occurs when aqueous solutions of anions (negatively charged ions) and cations (positively charged ions) combine to form a compound that is insoluble is known as precipitation. The insoluble solid is called the precipitate, and the remaining liquid is called the supernate. See Figure2.1
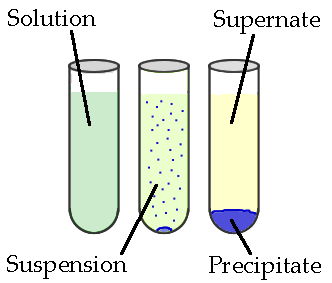
Real life example: The white precipitate formed by acid rain on a marble statue:
\[CaCO_3(aq)+H_2SO_4(aq) \rightarrow CaSO_4(s)+H_2O(l)+CO_2(g) \nonumber \]
An example of a precipitation reaction is the reaction between silver nitrate and sodium iodide. This reaction is represented by the chemical equation :
AgNO3 (aq)+ NaI (aq) → AgI (s) + NaNO3 (aq)
Since all of the above species are in aqueous solutions, they are written as ions, in the form:
Ag+ +NO3- (aq)+ Na+ (aq) + I- (aq) → AgI (s) + Na+ (aq) + NO3- (aq)
Ions that appear on both sides of the equation are called spectator ions. These ions do not affect the reaction and are removed from both sides of the equation to reveal the net ionic equation, as written below:
Ag+ (aq) + I- (aq) → AgI (s)
In this reaction, the solid, AgI, is known as the precipitate. The formation of a precipitate is one of the many indicators that a chemical reaction has taken place.
Acid-base, or neutralization reaction
A neutralization reaction occurs when an acid and base are mixed together. An acid is a substance that produces H+ ions in solution, whereas a base is a substance that that produces OH- ions in solution. A typical acid-base reaction will produce an ionic compound called a salt and water. A typical acid-base reaction is the reaction between hydrochloric acid and sodium hydroxide. This reaction is represented by the equation:
\[\ce{HCl (aq) + NaOH (aq) \rightarrow NaCl (aq)+ H_2O (l)} \nonumber \]
In this reaction, \(HCl\) is the acid, \(NaOH\) is the base, and \(NaCl\) is the salt. Real life example: Baking soda reacts with vinegar is a neutralization reaction.
Video: Vinegar and Baking Soda Reaction with Explanation
Oxidation-Reduction (Redox) Reactions
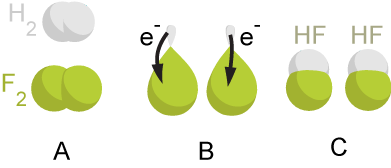
A redox reaction occurs when the oxidation number of atoms involved in the reaction are changed. Oxidation is the process by which an atom’s oxidation number is increased, and reduction is the process by which an atom’s oxidation number is decreased. If the oxidation states of any elements in a reaction change, the reaction is an oxidation-reduction reaction. An atom that undergoes oxidation is called the reducing agent, and the atom that undergoes reduction is called the oxidizing agent. An example of a redox reaction is the reaction between hydrogen gas and fluorine gas:
\[H_2 (g) + F_2 (g) \rightarrow 2HF (g) \label{redox1} \]
In this reaction, hydrogen is oxidized from an oxidation state of 0 to +1, and is thus the reducing agent. Fluorine is reduced from 0 to -1, and is thus the oxidizing agent.
Real life example: The cut surface of an apple turns brownish after exposed to the air for a while.
Video: Why Do Apples Turn Brown?
Combustion Reaction
A combustion reaction is a type of redox reaction during which a fuel reacts with an oxidizing agent, resulting in the release of energy as heat. Such reactions are exothermic, meaning that energy is given off during the reaction. An endothermic reaction is one which absorbs heat. A typical combustion reaction has a hydrocarbon as the fuel source, and oxygen gas as the oxidizing agent. The products in such a reaction would be \(CO_2\) and \(H_2O\).
\[C_xH_yO_z+O_2 \rightarrow CO_2+H_2O \;\;\; \text{(unbalanced)} \nonumber \]
Such a reaction would be the combustion of glucose in the following equation
\[C_6H_{12}O_6 (s) + 6O_2 (g) \rightarrow 6CO_2 (g) + 6H_2O (g) \nonumber \]
Real life example: explosion; burning.
Video: Combustion reactions come in many varieties. Here's a collection of various examples, all of which require oxygen, activation energy, and of course, fuel
Synthesis Reactions
A synthesis reaction occurs when one or more compounds combines to form a complex compound. The simplest equation of synthesis reaction is illustrated below.
.png?revision=2)
An example of such a reaction is the reaction of silver with oxygen gas to form silver oxide:
\[2Ag (s) +O_2 (g) \rightarrow 2AgO (s) \nonumber \]
Real life example: Hydrogen gas is burned in air (reacts with oxygen) to form water:
\[2H_2(g) + O_2(g) \rightarrow 2H_2O(l) \nonumber \]
Decomposition Reactions
A decomposition reaction is the opposite of a synthesis reaction. During a decomposition reaction, a more complex compound breaks down into multiple simpler compounds. A classic example of this type of reaction is the decomposition of hydrogen peroxide into oxygen and hydrogen gas:
\[H_2O_2 (l) \rightarrow H_2 (g) + O_2 (g) \nonumber \]

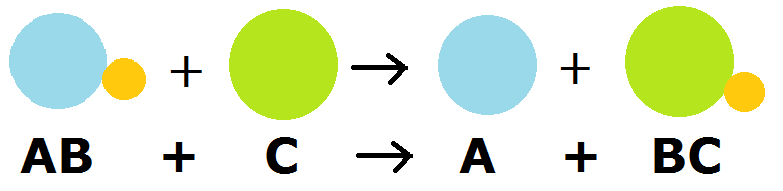
Single Replacement Reactions
A type of oxidation-reduction reaction in which an element in a compound is replaced by another element.
.png?revision=2)
An example of such a reaction is:
\[Cu (s) + AgNO_3 (aq) \rightarrow Ag(s) + Cu(NO_3)_2 (aq) \nonumber \]
This is also a redox reaction.
Problems
1) C3H6O3 + O2 → CO2 (g) +H2O (g)
a) What type of reaction is this?
b) Is is exothermic or endothermic? Explain.
2) Given the oxidation-reduction reaction :
Fe (s) + CuSO4 (aq)→ FeSO4 (aq)+ Cu (s)
a) Which element is the oxidizing agent and which is the reducing agent?
b) How do the oxidation states of these species change?
3) Given the equation:
AgNO3 (aq) + KBr (aq) → AgBr (s) +KNO3 (aq)
a) What is the net ionic reaction?
b) Which species are spectator ions?
4) 2 HNO3 (aq) + Sr(OH)2 (aq) → Sr(NO3)2 (aq) +2 H2O (l)
a) In this reaction, which species is the acid and which is the base?
b) Which species is the salt?
c) If 2 moles of HNO3 and 1 mole of Sr(OH)2 are used, resulting in 0.85 moles of Sr(NO3)2 , what is the percent yield (with respect to moles) of Sr(NO3)2 ?
5) Identify the type of the following reactions:
a) Al(OH)3 (aq) + HCl (aq) → AlCl3 (aq) + H2O (l)
b) MnO2 + 4H+ + 2Cl- → Mn2+ + 2H2O (l) + Cl2 (g)
c) P4 (s) + Cl2 (g) → PCl3 (l)
d) Ca (s) + 2H2O (l) → Ca(OH)2 (aq) + H2 (g)
e) AgNO3 (aq) + NaCl (aq) → AgCl (s) + NaNO3 (aq)
Solutions
1a) It is a combustion reaction
1b) It is exothermic, because combustion reactions give off heat
2a) Cu is the oxidizing agent and Fe is the reducing agent
2b) Fe changes from 0 to +2, and Cu changes from +2 to 0.
3a) Ag+ (aq) + Br- (aq) → AgBr (s)
3b) The spectator ions are K+ and NO3-
4a) HNO3 is the acid and Sr(OH)2 is the base
4b) Sr(NO3)2 is the salt
4c) According to the stoichiometric coefficients, the theoretical yield of Sr(NO3)2 is one mole. The actual yield was 0.85 moles. Therefore the percent yield is:
(0.85/1.0) * 100% = 85%
5a) Acid-base
5b) Oxidation-reduction
5c) Synthesis
5d) Single-replacement reaction
5e) Double replacement reaction
References
- Petrucci, Ralph H., William S. Harwood, F. G. Herring, and Jeffry D. Madura. General Chemistry. 9th ed. Upper Saddle River: Prentice Education Inc, 2007, Print.
Contributors and Attributions
- Priya Muley

